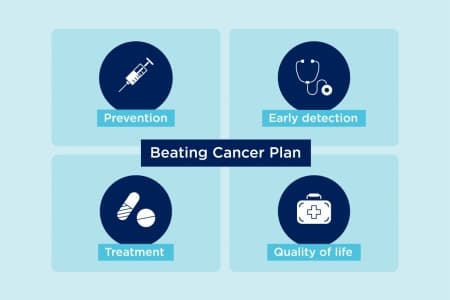
With this ambitious plan, the EU has devised a four-pillar approach to overcome cancer as a leading cause of death. These are:
prevention;
early detection;
diagnosis and treatment; and
quality of life of cancer patients and survivors.
AmCham EU shares the European Commission’s ambition to take on cancer. A holistic approach that encompasses the entire disease pathway, while engaging the full arsenal of the life sciences sector in the fight, can help to improve the survival chances of cancer patients, while reducing the number of cases.
Check out our explainer of Europe’s Beating Cancer Plan.
:focal())
:focal())